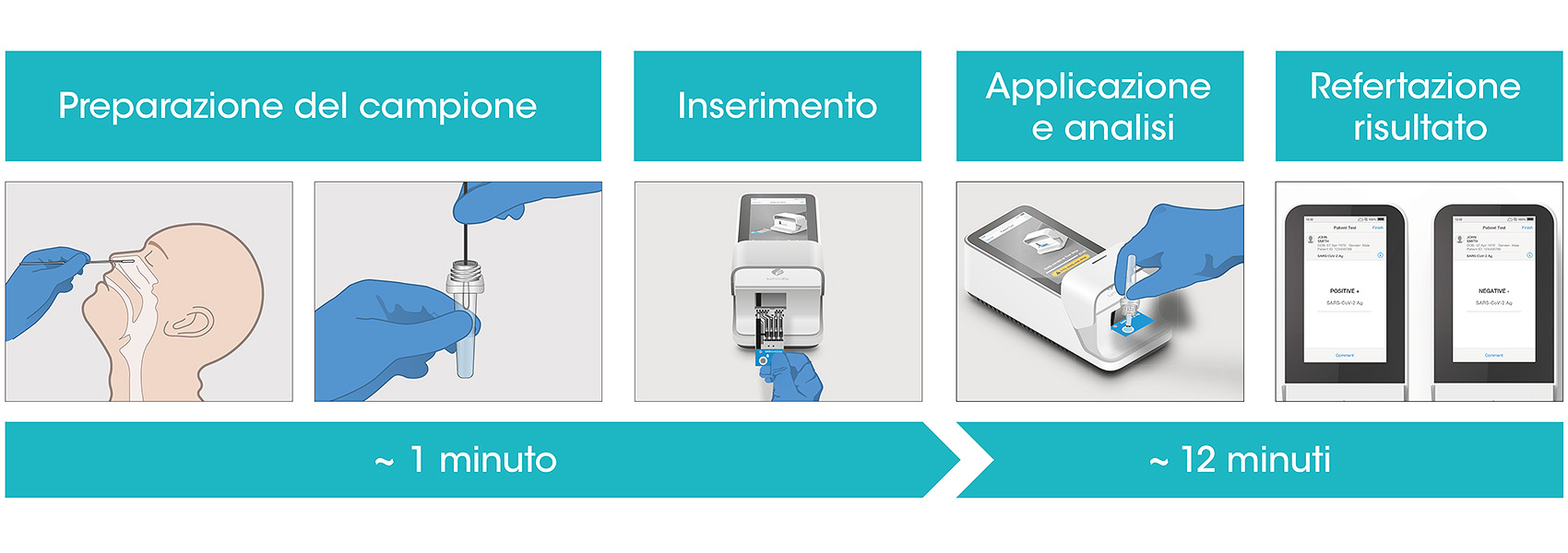
Test Covid 19 Ultima Generazione, Immunofluorescenza con lettura microfluidica - Farmacia Pescetto di Pegli, Genova

Test Covid 19 Ultima Generazione, Immunofluorescenza con lettura microfluidica - Farmacia Pescetto di Pegli, Genova

LumiraDx Expands its Cardiovascular Testing Portfolio with CE Marking of its NT-proBNP Test and new Exclusion Claim for its D-Dimer Test
LumiraDx launches C-Reactive Protein test to combat antimicrobial resistance in India, ET HealthWorld

A rapid, high-sensitivity SARS-CoV-2 nucleocapsid immunoassay to aid diagnosis of acute COVID-19 at the point of care | medRxiv

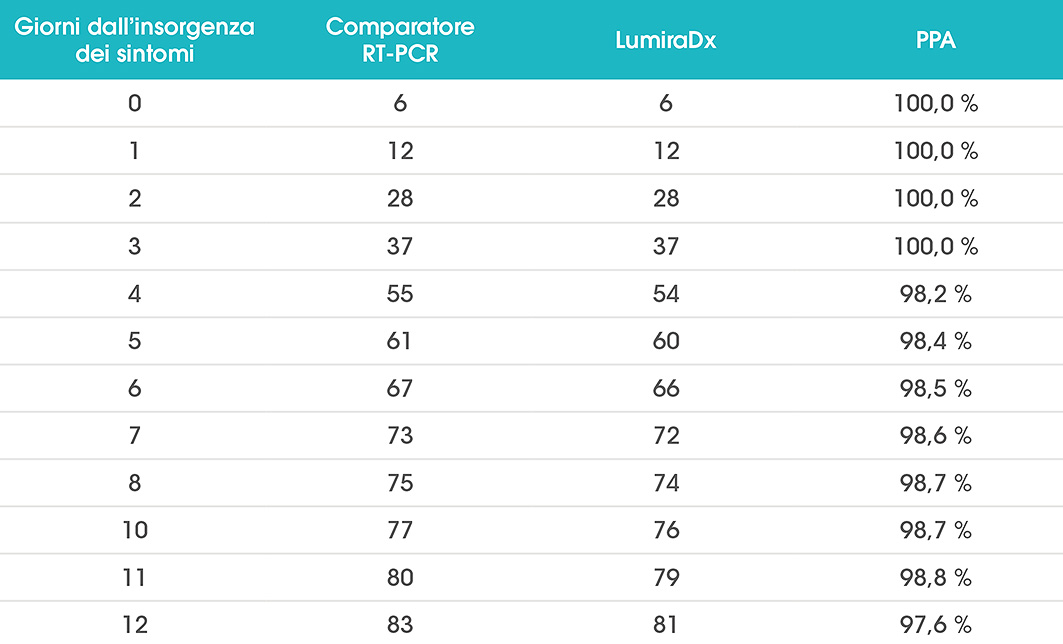
LumiraDx riceve l'autorizzazione per il test antigenico SARS-CoV-2 in Giappone e Brasile; l'Italia raccomanda l'espansione del test antigenico basato su microfluidica